By Ivan Morgunov, CEO of Unlimited Bio
Summary
I’m running an N-of-1 “split-scalp” experiment: one half of my scalp receives local VEGF gene therapy, the other half receives placebo, so each side serves as the other’s control.
The biological bet is that VEGF—the main angiogenesis driver—can change the microvascular environment around hair follicles enough to produce a measurable hair signal in real-world conditions.
Because hair growth is slow and noisy, we lean on repeatable measurement (imaging, phototrichograms, and marked scalp sites).
This article is informational. I’m describing a self-experiment and the reasoning behind its design. It is not medical advice. The intervention is described as gene therapy in the broad sense—delivery of genetic material to drive the production of a protein in a targeted tissue—and that category carries real uncertainty.
Introduction
Hair grows slowly. Meanwhile my scalp—and my ability to photograph it consistently—changes with seasons, stress, sleep, grooming habits, illness, medications, and lighting. If I simply compare “before vs. after,” after getting a certain treatment, I can easily end up measuring noise. That’s why I’m treating this like a protocol-driven test: define what I’ll measure, how I’ll measure it, when I’ll measure it, and what would count as a meaningful change.
The split-scalp design adds one more layer of discipline: the control is built into the same head, the same life, the same month. If the outcome is negative, it’s still valuable because it’s negative in a way that’s interpretable.
Why do this?
In androgenetic alopecia, the core visible process is follicle miniaturization: terminal hairs gradually become thinner and shorter, and growth cycles shorten until the hair that remains is barely noticeable. The pattern tends to be region-specific because follicle sensitivity to androgens varies across the scalp.
VEGF (vascular endothelial growth factor) is one of the core signals that drives angiogenesis—growth and remodeling of small blood vessels. Hair follicles aren’t just “mini-organs of keratin.” In active growth (anagen), the follicle expands and its vascular support changes; when follicles regress (catagen), that support also shifts.
In animal work, increasing VEGF in skin strengthened perifollicular vasculature and accelerated hair regrowth, while blocking VEGF slowed hair growth. A key detail in that line of evidence is that the effect can disappear in settings where vascular support is absent—an argument that the mechanism is meaningfully “vascular,” not merely cosmetic correlation.
Human evidence is less direct, but suggestive: small studies have reported differences in scalp microcirculation and oxygenation in balding regions compared with controls. For example, subcutaneous blood flow in early male-pattern baldness was reported lower than in matched controls (13.7 ± 9.6 vs 35.7 ± 10.5 ml/100 g/min; 14 per group), and transcutaneous oxygen measurements over balding areas have been reported lower than controls in a small cohort (9 per group). More recent work using high-frequency ultrasound has also described scalp-parameter differences between AGA and healthy volunteers.
Those findings do not prove that vascular change is the root cause—it may be downstream remodeling, parallel remodeling, or a partial contributor. The point of my experiment is narrower: if I locally push a VEGF signal, do I see any measurable “hair signal,” side-by-side, on the same person?
In broad terms, gene therapy is the delivery of genetic material designed to change what a cell produces. One recognized platform is plasmid DNA—circular DNA engineered to carry a gene into human cells. In my protocol, the active material is a plasmid gene therapy delivered locally to scalp skin via multiple microinjections, mirrored by placebo microinjections on the other side.
Summary
I’m running an N-of-1 “split-scalp” experiment: one half of my scalp receives local VEGF gene therapy, the other half receives placebo, so each side serves as the other’s control.
The biological bet is that VEGF—the main angiogenesis driver—can change the microvascular environment around hair follicles enough to produce a measurable hair signal in real-world conditions.
Because hair growth is slow and noisy, we lean on repeatable measurement (imaging, phototrichograms, and marked scalp sites).
This article is informational. I’m describing a self-experiment and the reasoning behind its design. It is not medical advice. The intervention is described as gene therapy in the broad sense—delivery of genetic material to drive the production of a protein in a targeted tissue—and that category carries real uncertainty.
Introduction
Hair grows slowly. Meanwhile my scalp—and my ability to photograph it consistently—changes with seasons, stress, sleep, grooming habits, illness, medications, and lighting. If I simply compare “before vs. after,” after getting a certain treatment, I can easily end up measuring noise. That’s why I’m treating this like a protocol-driven test: define what I’ll measure, how I’ll measure it, when I’ll measure it, and what would count as a meaningful change.
The split-scalp design adds one more layer of discipline: the control is built into the same head, the same life, the same month. If the outcome is negative, it’s still valuable because it’s negative in a way that’s interpretable.
Why do this?
In androgenetic alopecia, the core visible process is follicle miniaturization: terminal hairs gradually become thinner and shorter, and growth cycles shorten until the hair that remains is barely noticeable. The pattern tends to be region-specific because follicle sensitivity to androgens varies across the scalp.
VEGF (vascular endothelial growth factor) is one of the core signals that drives angiogenesis—growth and remodeling of small blood vessels. Hair follicles aren’t just “mini-organs of keratin.” In active growth (anagen), the follicle expands and its vascular support changes; when follicles regress (catagen), that support also shifts.
In animal work, increasing VEGF in skin strengthened perifollicular vasculature and accelerated hair regrowth, while blocking VEGF slowed hair growth. A key detail in that line of evidence is that the effect can disappear in settings where vascular support is absent—an argument that the mechanism is meaningfully “vascular,” not merely cosmetic correlation.
Human evidence is less direct, but suggestive: small studies have reported differences in scalp microcirculation and oxygenation in balding regions compared with controls. For example, subcutaneous blood flow in early male-pattern baldness was reported lower than in matched controls (13.7 ± 9.6 vs 35.7 ± 10.5 ml/100 g/min; 14 per group), and transcutaneous oxygen measurements over balding areas have been reported lower than controls in a small cohort (9 per group). More recent work using high-frequency ultrasound has also described scalp-parameter differences between AGA and healthy volunteers.
Those findings do not prove that vascular change is the root cause—it may be downstream remodeling, parallel remodeling, or a partial contributor. The point of my experiment is narrower: if I locally push a VEGF signal, do I see any measurable “hair signal,” side-by-side, on the same person?
In broad terms, gene therapy is the delivery of genetic material designed to change what a cell produces. One recognized platform is plasmid DNA—circular DNA engineered to carry a gene into human cells. In my protocol, the active material is a plasmid gene therapy delivered locally to scalp skin via multiple microinjections, mirrored by placebo microinjections on the other side.
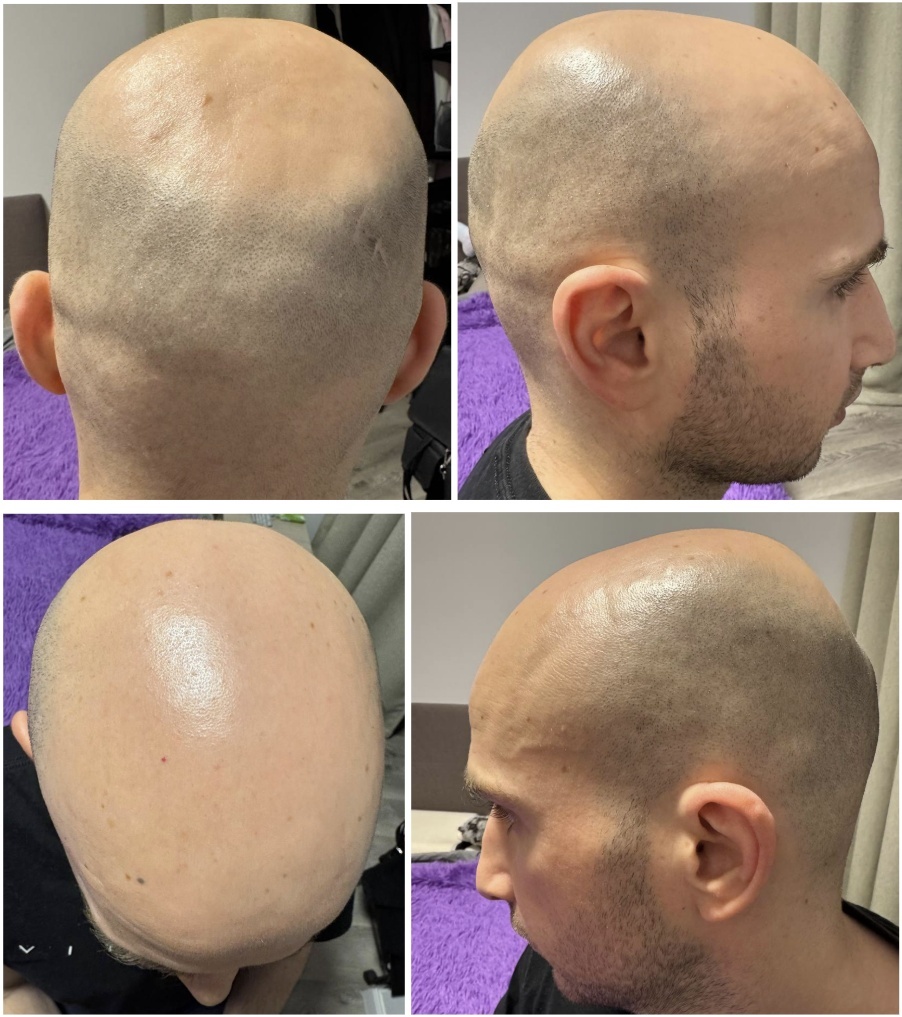
Photographs Before the Injections and Hair Transplant
A side note on Minoxydil
Minoxidil, the most popular hair loss medication, often enters the VEGF discussion for two reasons. First, some studies have reported a rapid increase in blood flow in balding scalp after topical minoxidil, while others reported no measurable blood-flow change—so the vascular story is not clean or uniform.
Second, cell-model evidence suggests minoxidil can upregulate VEGF expression in human dermal papilla cells, providing at least a plausible mechanistic bridge between “hair drugs” and “vascular niche.”
None of that proves what causes androgenetic alopecia. It does explain why a VEGF hypothesis doesn’t sound outlandish on its face: follicle performance depends on its microenvironment, and vasculature is part of that environment.
Preliminary hair transplant
At later stages of androgenetic alopecia, some areas of the scalp have been without terminal hairs for a long time. Even if I improve the tissue environment (inflammation, perfusion, “niche support”), that does not guarantee new functional follicles will reappear. In that context, hair transplantation remains the primary intervention that can reintroduce follicles, albeit within clear limits and constraints.
In this project I completed a first transplantation stage (about 2,700 grafts into the recipient zone). For the experiment, that matters because it creates multiple biological contexts on the same scalp:
If VEGF modulation produces an effect, it may show up differently across those contexts—and the protocol explicitly tries to capture that rather than blur it into a “single number”.
Minoxidil, the most popular hair loss medication, often enters the VEGF discussion for two reasons. First, some studies have reported a rapid increase in blood flow in balding scalp after topical minoxidil, while others reported no measurable blood-flow change—so the vascular story is not clean or uniform.
Second, cell-model evidence suggests minoxidil can upregulate VEGF expression in human dermal papilla cells, providing at least a plausible mechanistic bridge between “hair drugs” and “vascular niche.”
None of that proves what causes androgenetic alopecia. It does explain why a VEGF hypothesis doesn’t sound outlandish on its face: follicle performance depends on its microenvironment, and vasculature is part of that environment.
Preliminary hair transplant
At later stages of androgenetic alopecia, some areas of the scalp have been without terminal hairs for a long time. Even if I improve the tissue environment (inflammation, perfusion, “niche support”), that does not guarantee new functional follicles will reappear. In that context, hair transplantation remains the primary intervention that can reintroduce follicles, albeit within clear limits and constraints.
In this project I completed a first transplantation stage (about 2,700 grafts into the recipient zone). For the experiment, that matters because it creates multiple biological contexts on the same scalp:
- a donor area that is healing,
- a recipient area where grafts are revascularizing and maturing,
- areas with preserved native hair,
- long-bald areas.
If VEGF modulation produces an effect, it may show up differently across those contexts—and the protocol explicitly tries to capture that rather than blur it into a “single number”.
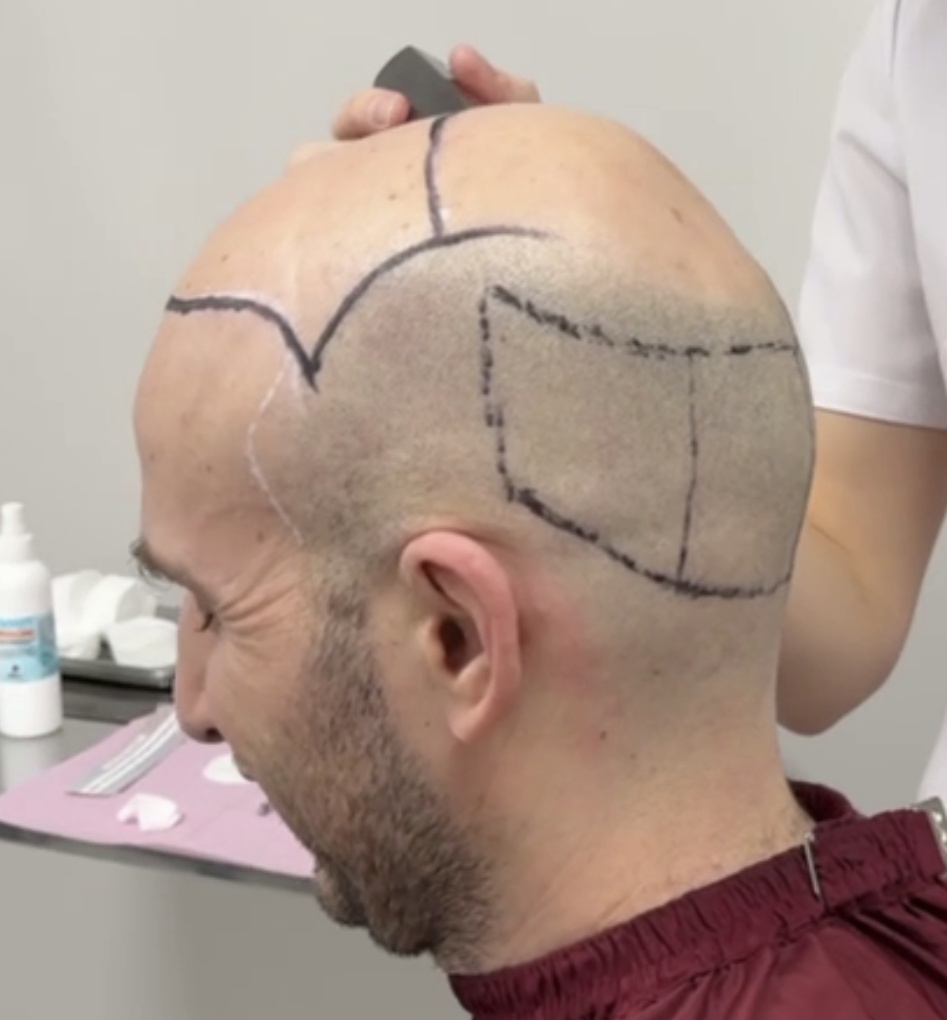
Hair Transplant Zone and Donor Zone Marked
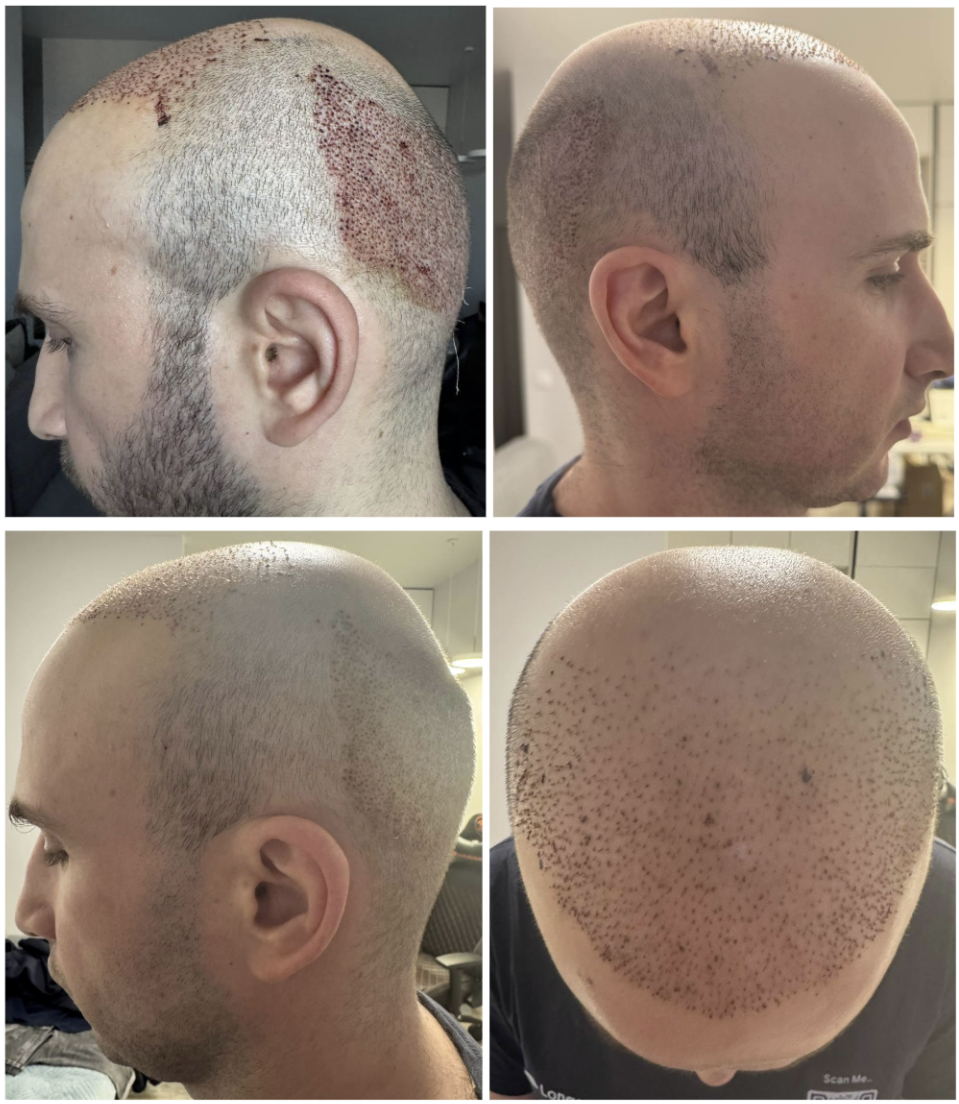
Photographs After the Hair Transplantation
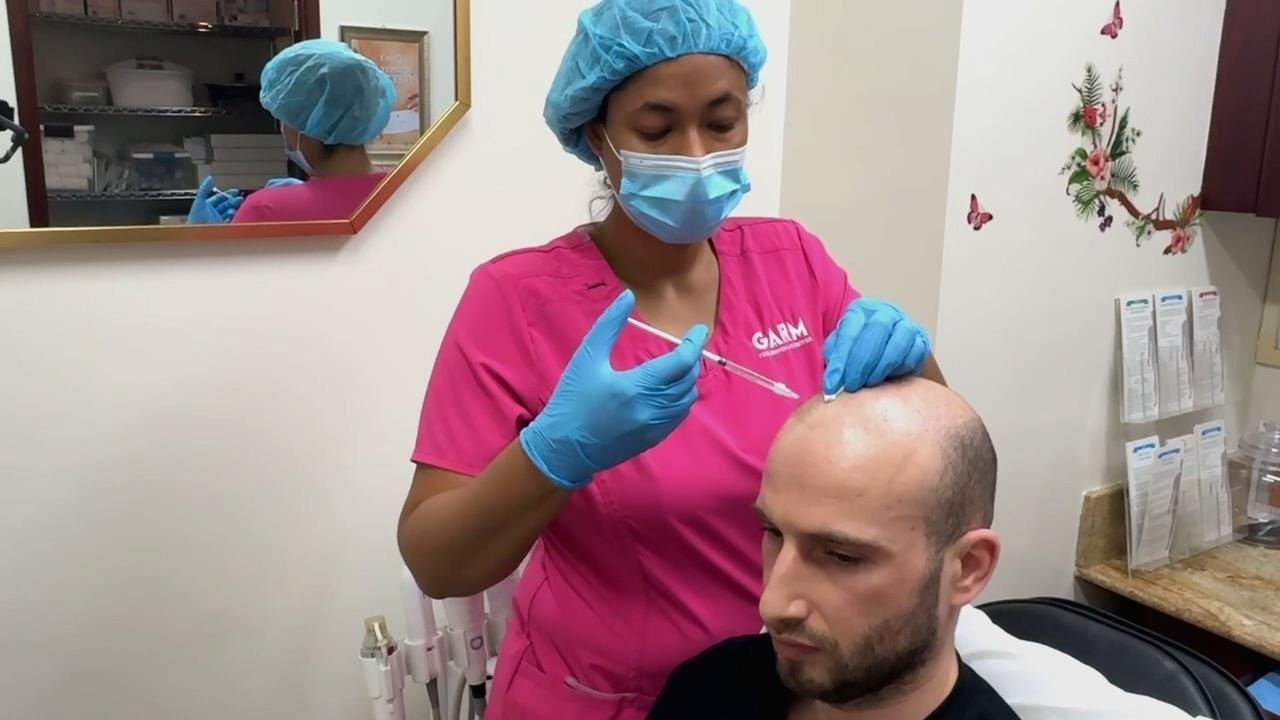
Administration Procedure
Before any injections, I lock down baseline measurements with standardized imaging (photo/video dermoscopy), phototrichograms, and capillaroscopy. We use persistent scalp marks so that follow-up measurements sample the same micro-areas.
Then we randomize which half receives the active intervention and which half receives placebo. Both halves undergo symmetric microinjection procedures using a pre-defined map. Follow-up measurements are taken on a schedule (not specified in the provided article text) and I compare change over time between the two sides, within each scalp zone (donor/recipient/intact hair-bearing/long-bald). At all stages of this experiment during the whole duration, both the doctor carrying out the injections and me are blinded.
The donor zone behaves like a controlled wound: healing, inflammation, and vascular remodeling are strong signals on their own. The recipient zone is a different biological story, written by revascularization and graft maturation. Intact hair-bearing scalp is more stable tissue. Long-bald areas may have lost follicular functionality, so “better environment” doesn’t necessarily translate to terminal hairs. Treating those contexts separately is an attempt to keep interpretation honest.
Video of the procedure is available on YouTube.
Before any injections, I lock down baseline measurements with standardized imaging (photo/video dermoscopy), phototrichograms, and capillaroscopy. We use persistent scalp marks so that follow-up measurements sample the same micro-areas.
Then we randomize which half receives the active intervention and which half receives placebo. Both halves undergo symmetric microinjection procedures using a pre-defined map. Follow-up measurements are taken on a schedule (not specified in the provided article text) and I compare change over time between the two sides, within each scalp zone (donor/recipient/intact hair-bearing/long-bald). At all stages of this experiment during the whole duration, both the doctor carrying out the injections and me are blinded.
The donor zone behaves like a controlled wound: healing, inflammation, and vascular remodeling are strong signals on their own. The recipient zone is a different biological story, written by revascularization and graft maturation. Intact hair-bearing scalp is more stable tissue. Long-bald areas may have lost follicular functionality, so “better environment” doesn’t necessarily translate to terminal hairs. Treating those contexts separately is an attempt to keep interpretation honest.
Video of the procedure is available on YouTube.
Injection Procedure in GARM Clinic
A measurement example: phototrichograms
A phototrichogram is a magnified image of a small scalp area that enables counting hairs and estimating traits like density and the fraction of thin/miniaturized hairs. Marking the measurement sites (for example, with small tattoos) is one known strategy used in hair studies to improve repeatability of hair counts.
A phototrichogram is a magnified image of a small scalp area that enables counting hairs and estimating traits like density and the fraction of thin/miniaturized hairs. Marking the measurement sites (for example, with small tattoos) is one known strategy used in hair studies to improve repeatability of hair counts.
Limitations
Even with a “half-head control,” false signals are possible:
Even with a “half-head control,” false signals are possible:
- Spillover: the scalp halves are not isolated systems; systemic signals can blur differences.
- “Active placebo”: the very series of microinjections is not a neutral control. The microtrauma and short-lived inflammation can alter the skin and may even affect hair, triggering extracellular matrix remodeling and angiogenesis on their own. In trichology, a similar principle is used in microneedling.
- Baseline asymmetry: left and right may differ before I start, and transplant markings can introduce asymmetries—hence the emphasis on true pre-injection baselines.
- Hair-cycle timing and seasonality: follicles naturally cycle; specific timepoints can look like “effects.”
- Too many endpoints: if I measure many things, I can always find one that “looks good.” Pre-specifying primary vs secondary endpoints is how trials reduce that temptation.
- Background care: changes in medication, hair care, or additional procedures can contaminate interpretation.
Photographs after the injections
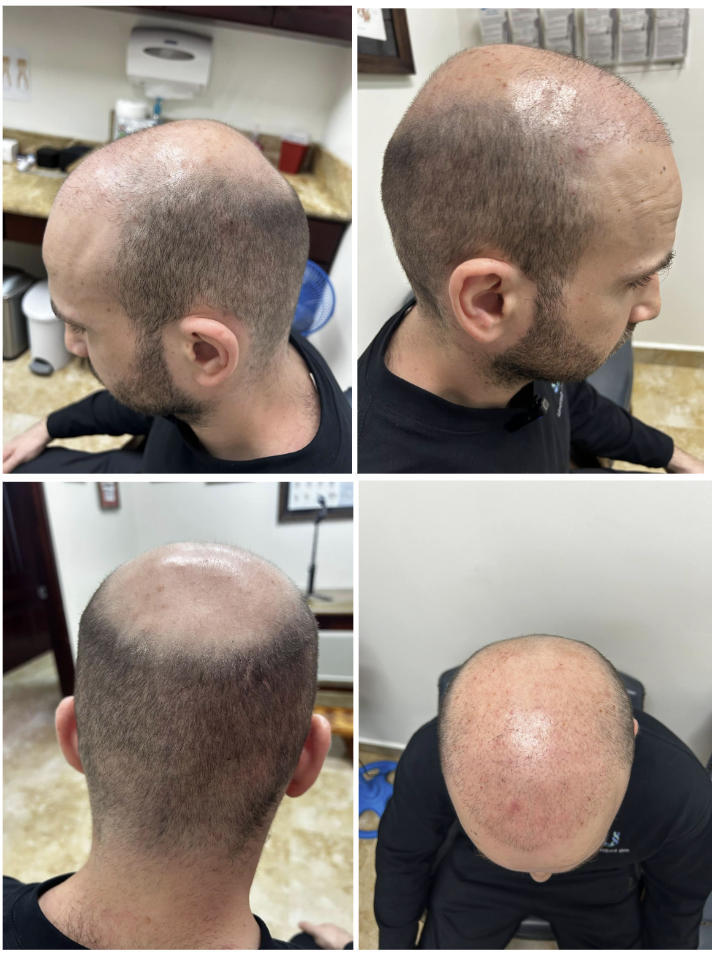
Conclusion
I’m looking for a coherent pattern: repeatable measurement sites, consistent directional differences between sides, and signals that make biological sense within each zone. Let’s see whether I’ll be able to acquire it.
Stay tuned for the 6-month update!
References
1. Richards JB, Yuan X, Geller F, et al. Male-pattern baldness susceptibility locus at 20p11. Nat Genet. 2008;40(11):1282–1284. doi:10.1038/ng.255. PubMed: https://pubmed.ncbi.nlm.nih.gov/18849991/
2. Yano K, Brown LF, Detmar M. Control of hair growth and follicle size by VEGF-mediated angiogenesis. J Clin Invest. 2001;107(4):409–417. doi:10.1172/JCI11317. PubMed: https://pubmed.ncbi.nlm.nih.gov/11181640/
3. Klemp P, Peters K, Hansted B. Subcutaneous blood flow in early male pattern baldness. J Invest Dermatol. 1989;92(5):725–726. doi:10.1111/1523-1747.ep12721603. PubMed: https://pubmed.ncbi.nlm.nih.gov/2715645/
4. Goldman BE, Fisher DM, Ringler SL. Transcutaneous PO2 of the scalp in male pattern baldness: a new piece to the puzzle. Plast Reconstr Surg. 1996;97(6):1109–1116. doi:10.1097/00006534-199605000-00003. PubMed: https://pubmed.ncbi.nlm.nih.gov/8628793/
5. Li L, et al. High-frequency ultrasonography of the scalp: A comparison between androgenetic alopecia and healthy volunteers. Skin Res Technol. 2024;30(8):e13863. doi:10.1111/srt.13863. PubMed: https://pubmed.ncbi.nlm.nih.gov/39081105/ | Full text: https://pmc.ncbi.nlm.nih.gov/articles/PMC11289427/
6. Wester RC, Maibach HI, Guy RH, Novak E. Minoxidil stimulates cutaneous blood flow in human balding scalps: pharmacodynamics measured by laser Doppler velocimetry and photopulse plethysmography. J Invest Dermatol. 1984;82(5):515–517. doi:10.1111/1523-1747.ep12261084. PubMed: https://pubmed.ncbi.nlm.nih.gov/6239893/
7. de Boer EM, Bezemer PD, Bruynzeel DP, Nieboer C. Does topical minoxidil increase skin blood flow? A laser Doppler flowmetry study. Acta Derm Venereol. 1988;68(3):271–274. PubMed: https://pubmed.ncbi.nlm.nih.gov/2455426/
8. Lachgar S, Charveron M, Gall Y, Bonafe JL. Minoxidil upregulates the expression of vascular endothelial growth factor in human hair dermal papilla cells. Br J Dermatol. 1998;138(3):407–411. doi:10.1046/j.1365-2133.1998.02115.x. PubMed: https://pubmed.ncbi.nlm.nih.gov/9580790/
9. Venkataram A, Mysore V. Logic of Hair Transplantation. J Cutan Aesthet Surg. 2018;11(4):169–172. doi:10.4103/JCAS.JCAS_183_18. PubMed: https://pubmed.ncbi.nlm.nih.gov/30886469/ | Full text: https://pmc.ncbi.nlm.nih.gov/articles/PMC6371726/
10. Dhurat R, Sukesh M, Avhad G, Dandale A, Pal A, Pund P. A randomized evaluator blinded study of effect of microneedling in androgenetic alopecia: a pilot study. Int J Trichology. 2013;5(1):6–11. doi:10.4103/0974-7753.114700. PubMed: https://pubmed.ncbi.nlm.nih.gov/23960389/ | Full text: https://pmc.ncbi.nlm.nih.gov/articles/PMC3746236/
I’m looking for a coherent pattern: repeatable measurement sites, consistent directional differences between sides, and signals that make biological sense within each zone. Let’s see whether I’ll be able to acquire it.
Stay tuned for the 6-month update!
References
1. Richards JB, Yuan X, Geller F, et al. Male-pattern baldness susceptibility locus at 20p11. Nat Genet. 2008;40(11):1282–1284. doi:10.1038/ng.255. PubMed: https://pubmed.ncbi.nlm.nih.gov/18849991/
2. Yano K, Brown LF, Detmar M. Control of hair growth and follicle size by VEGF-mediated angiogenesis. J Clin Invest. 2001;107(4):409–417. doi:10.1172/JCI11317. PubMed: https://pubmed.ncbi.nlm.nih.gov/11181640/
3. Klemp P, Peters K, Hansted B. Subcutaneous blood flow in early male pattern baldness. J Invest Dermatol. 1989;92(5):725–726. doi:10.1111/1523-1747.ep12721603. PubMed: https://pubmed.ncbi.nlm.nih.gov/2715645/
4. Goldman BE, Fisher DM, Ringler SL. Transcutaneous PO2 of the scalp in male pattern baldness: a new piece to the puzzle. Plast Reconstr Surg. 1996;97(6):1109–1116. doi:10.1097/00006534-199605000-00003. PubMed: https://pubmed.ncbi.nlm.nih.gov/8628793/
5. Li L, et al. High-frequency ultrasonography of the scalp: A comparison between androgenetic alopecia and healthy volunteers. Skin Res Technol. 2024;30(8):e13863. doi:10.1111/srt.13863. PubMed: https://pubmed.ncbi.nlm.nih.gov/39081105/ | Full text: https://pmc.ncbi.nlm.nih.gov/articles/PMC11289427/
6. Wester RC, Maibach HI, Guy RH, Novak E. Minoxidil stimulates cutaneous blood flow in human balding scalps: pharmacodynamics measured by laser Doppler velocimetry and photopulse plethysmography. J Invest Dermatol. 1984;82(5):515–517. doi:10.1111/1523-1747.ep12261084. PubMed: https://pubmed.ncbi.nlm.nih.gov/6239893/
7. de Boer EM, Bezemer PD, Bruynzeel DP, Nieboer C. Does topical minoxidil increase skin blood flow? A laser Doppler flowmetry study. Acta Derm Venereol. 1988;68(3):271–274. PubMed: https://pubmed.ncbi.nlm.nih.gov/2455426/
8. Lachgar S, Charveron M, Gall Y, Bonafe JL. Minoxidil upregulates the expression of vascular endothelial growth factor in human hair dermal papilla cells. Br J Dermatol. 1998;138(3):407–411. doi:10.1046/j.1365-2133.1998.02115.x. PubMed: https://pubmed.ncbi.nlm.nih.gov/9580790/
9. Venkataram A, Mysore V. Logic of Hair Transplantation. J Cutan Aesthet Surg. 2018;11(4):169–172. doi:10.4103/JCAS.JCAS_183_18. PubMed: https://pubmed.ncbi.nlm.nih.gov/30886469/ | Full text: https://pmc.ncbi.nlm.nih.gov/articles/PMC6371726/
10. Dhurat R, Sukesh M, Avhad G, Dandale A, Pal A, Pund P. A randomized evaluator blinded study of effect of microneedling in androgenetic alopecia: a pilot study. Int J Trichology. 2013;5(1):6–11. doi:10.4103/0974-7753.114700. PubMed: https://pubmed.ncbi.nlm.nih.gov/23960389/ | Full text: https://pmc.ncbi.nlm.nih.gov/articles/PMC3746236/
